Hydrogen Liquefier
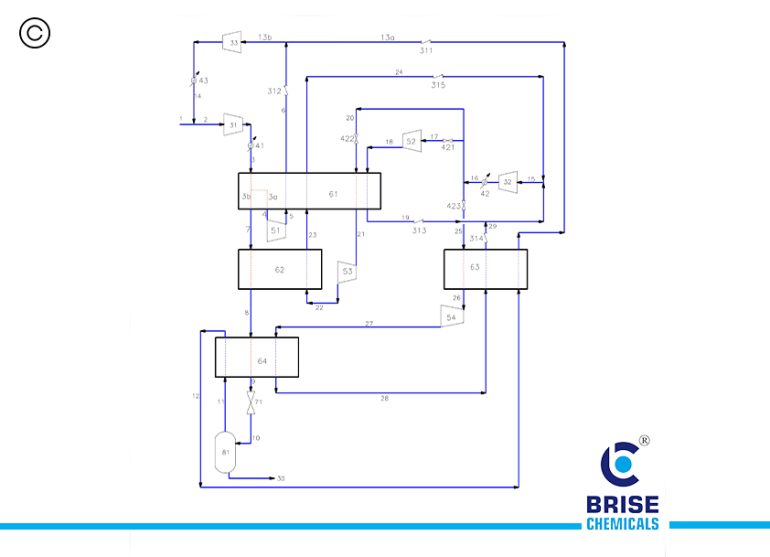
Transportation and storage of hydrogen are critical to its large-scale adoption and hence, hydrogen is most commonly transported and delivered as a liquid when high-volume transport is needed in the absence of pipelines therefore, liquefaction and storage processes for LH2 must, however, be both safe and efficient for liquid hydrogen to be viable as an energy carrier.
Identifying the most promising liquefaction processes and associated transport and storage technologies is therefore crucial. Gaseous hydrogen is liquefied by cooling it to below −253°C (20°K). The Ideal liquefaction work required to turn hydrogen gas into liquid (energy) is thermodynamically determined by the state quantity at the start and the end of the liquefaction process. Assuming a starting point of.
Atmospheric pressure and 303 K and an endpoint of saturated liquid hydrogen at atmospheric pressure, the ideal liquefaction work is approximately 3.90 kWh/kg (0.35 kWh/Nm3 ). The conventionally available liquefaction methods require substantially higher energies typically in the range of 9-13 kWh/kg liquid hydrogen for Claude and Brayton cycles, depending on the equipment efficiencies, cooling cycle, and size of the liquefaction operation. There is no such process available with the optimum equipment efficiencies and power consumption approaching the value of 3.90 kWh/kg.
In view of the problem, BRISE CHEMICALS had developed its own unique and patented process for hydrogen liquefaction to serve various industries and the environment as a whole. This unique Liquefaction of hydrogen opens up new avenues for energy storage, transportation, and utilization, bringing Humanity one step closer to a sustainable and Green future.
Features of Brise Chemicals H2 Liquefier
- Patent Process and product
- Lowest power consumption
- Only H2 as a refrigerant
- Less capital expenses
- Smaller Foot print

